South Africa will not be rushed into launching the long-acting HIV prevention injection lenacapavir, according to Health Minister Dr. Aaron Motsoaledi, who addressed the issue during a private media briefing held alongside a Southern African Development Community (SADC) health ministers’ meeting.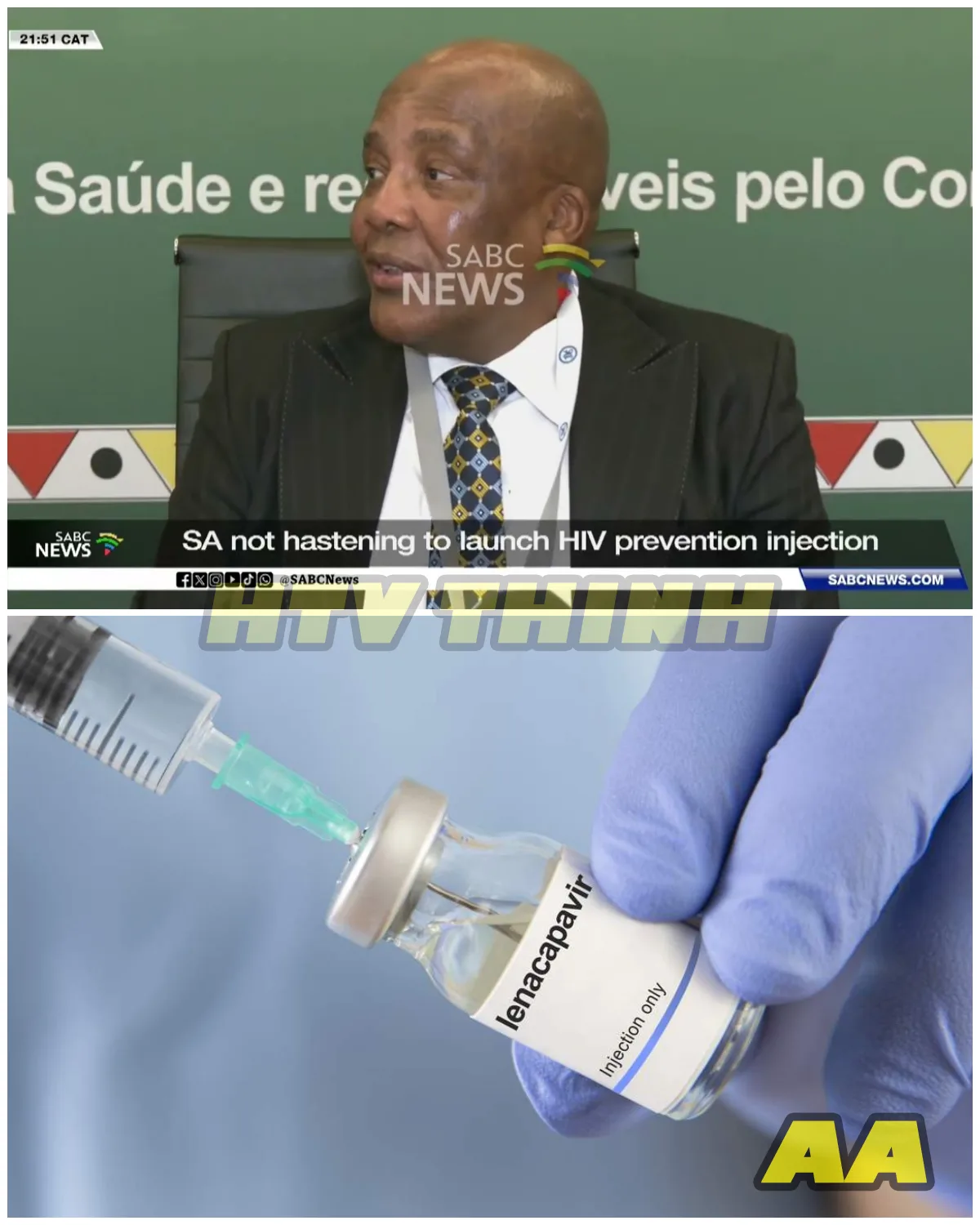
His message was measured but firm: while other countries have already begun rolling out the drug, South Africa will proceed only when it is fully prepared.
The long-acting injection, often referred to as “lenapa” in public discussion, represents a significant advancement in HIV prevention.
Administered less frequently than daily oral pre-exposure prophylaxis (PrEP), it has the potential to transform prevention strategies, particularly in high-burden countries such as South Africa.
Given the country’s large HIV-positive population and the ongoing efforts to curb new infections, the anticipation around the injection has been substantial.
However, Dr. Motsoaledi made it clear that readiness—not speed—will determine the timeline.
He indicated that the launch could possibly take place by May, but emphasized that no official date has been confirmed.
Preparations are still underway, and the government intends to ensure that the rollout is structured, equitable, and adequately supplied.
In his remarks, the minister acknowledged that countries such as Zimbabwe and Kenya have already initiated their rollout processes.
Rather than framing their early launches as competition, he congratulated them for making a start.
At the same time, he stressed that each country must move at its own pace, based on its logistical readiness and healthcare infrastructure.

According to Dr. Motsoaledi, South Africa had resisted pressure to launch earlier, particularly during a period when the Global Fund was encouraging rollout to coincide with a replenishment event held shortly before the G20 heads of state meeting.
The minister suggested that launching with only a limited number of doses already in the country would not have been prudent.
In his view, initiating a national program without sufficient stock and distribution capacity could create unrealistic expectations and disrupt service delivery.
South Africa has been allocated 988,000 doses of the injection.
The government’s strategy is to wait until the allocated supply is fully available before launching the program nationally.
This approach is intended to avoid fragmented distribution and ensure that facilities can provide consistent access to patients once the rollout begins.
The health department plans to introduce the injection across approximately 350 health facilities nationwide.
Such an expansion requires careful coordination, training of healthcare personnel, community education, and supply chain management.
The minister emphasized that a responsible rollout demands more than symbolic announcements; it requires operational readiness at every level of the healthcare system.
The cautious approach reflects lessons learned from previous large-scale public health interventions.
South Africa’s experience with HIV treatment programs, antiretroviral therapy scale-up, and COVID-19 vaccination campaigns has underscored the importance of logistical preparation.
Stock shortages, uneven distribution, and communication gaps can undermine public trust and weaken program effectiveness.
By delaying the launch until supply and infrastructure align, the health department aims to avoid those pitfalls.
Dr. Motsoaledi’s stance suggests that political optics—such as launching during a high-profile international meeting—will not dictate the country’s public health decisions.
The broader context of the decision is significant.
South Africa carries one of the highest HIV burdens globally.
Prevention remains a cornerstone of the national HIV strategy, alongside testing, treatment, and viral suppression.
The introduction of a long-acting prevention injection could address adherence challenges associated with daily oral PrEP, particularly among populations at higher risk.

Yet introducing a new biomedical tool into an already complex health system requires careful integration.
Training healthcare workers on administration protocols, monitoring side effects, maintaining cold chain requirements, and ensuring patient follow-up are all essential components of a successful launch.
Dr. Motsoaledi’s remarks also underscore a philosophy of sovereign public health planning.
While international partners such as the Global Fund play a critical role in financing and supporting HIV programs, South Africa intends to determine the timing of its rollout based on domestic readiness assessments.
The allocation of 988,000 doses represents a substantial commitment.
However, distribution must be equitable across provinces and communities.
Selecting 350 facilities as initial rollout sites suggests a phased approach, likely targeting high-burden districts first.
Ensuring that these sites are adequately staffed and supplied is central to the strategy.
The minister’s statement that “each country must launch as they are ready” reflects an understanding that health systems differ in capacity, geography, and patient load.
Zimbabwe and Kenya may have begun earlier, but South Africa’s scale and complexity necessitate tailored planning.
Public expectations remain high.
Advocacy groups and public health experts have long called for rapid access to innovative prevention tools.
Balancing urgency with prudence is therefore a delicate task.
Delays can be interpreted as hesitancy, yet premature launches can create operational setbacks.
By potentially targeting May for the launch—while withholding a definitive date—the health department signals progress without committing prematurely.
This approach allows ongoing logistical arrangements to continue without external pressure dictating timelines.

The emphasis on preparation also suggests that the department seeks to avoid disparities in access.
Launching in only a few facilities with limited stock could create inequity, where some patients gain access while others wait indefinitely.
A synchronized rollout across 350 sites would provide broader geographic coverage from the outset.
In addition to supply considerations, community awareness campaigns will be essential.
Public understanding of how the injection works, who qualifies, and how frequently it is administered will influence uptake.
Misinformation or confusion could undermine the program’s impact.
The minister’s remarks illustrate a balancing act between innovation and implementation.
South Africa recognizes the transformative potential of long-acting HIV prevention but insists that infrastructure readiness must precede public announcement.

Ultimately, the cautious approach signals a commitment to sustainability.
A well-prepared rollout may generate fewer headlines initially, but it could result in greater long-term impact and trust in the system.
As neighboring countries move ahead, South Africa’s strategy will be closely watched.
If the planned May timeframe materializes with adequate supply and operational readiness, the country could deliver one of the largest coordinated introductions of long-acting HIV prevention globally.





